Adenosine
 Purine nucleosides, such as adenosine, are important intracellular molecules of low molecular weight that participate in a wide variety of biochemical processes and serve as monomeric precursors of RNA and DNA. In addition, nucleosides play important roles as extracellular signaling molecules. Although adenosine is constitutively present at low levels in the extracellular space, metabolically stressful conditions, such as those that occur during injury, ischemia, and inflammation dramatically increase its extracellular concentration. It is clear that adenosine, acting mostly at its receptors, is a powerful signaling molecule that participates in the regulation of a wide variety of physiological and pathophysiological processes.
Purine nucleosides, such as adenosine, are important intracellular molecules of low molecular weight that participate in a wide variety of biochemical processes and serve as monomeric precursors of RNA and DNA. In addition, nucleosides play important roles as extracellular signaling molecules. Although adenosine is constitutively present at low levels in the extracellular space, metabolically stressful conditions, such as those that occur during injury, ischemia, and inflammation dramatically increase its extracellular concentration. It is clear that adenosine, acting mostly at its receptors, is a powerful signaling molecule that participates in the regulation of a wide variety of physiological and pathophysiological processes.
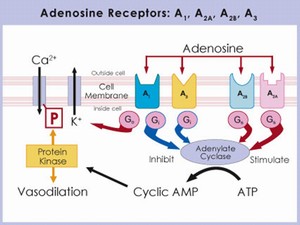
There are four subtypes of adenosine receptors (A1, A2a, A2b, A3) that detect local changes in adenosine levels. Adenosine receptors are members of the G-protein coupled receptor (GPCR) superfamily, and are typically thought to mediate stimulation or inhibition of adenylyl cyclase activity, and hence cyclic AMP levels. Receptor subtype selectivity is a critical aspect of adenosine therapeutics as each subtype has unique physiological effects (e.g. in the eye, A2a agonists increase IOP whereas A1 agonist reduce IOP).
Stimulation of the A1 receptor induces a range of biological responses including inhibition of nerve cell activities, depression of AV nodal conduction, lowering of intraocular pressure and global suppression of metabolic activity. Agonists of the A1 subtype can be used, therefore, as neuroprotective agents, as antinociceptive agents (especially when used in a direct intrathecal administration mode), as intravenous agents to stop supraventricular arrhythmias or to limit the ventricular response to supraventricular tachycardias (atrial fibrillation, atrial flutter, paroxysmal atrial tachycardia), and as intraocular pressure lowering agents.